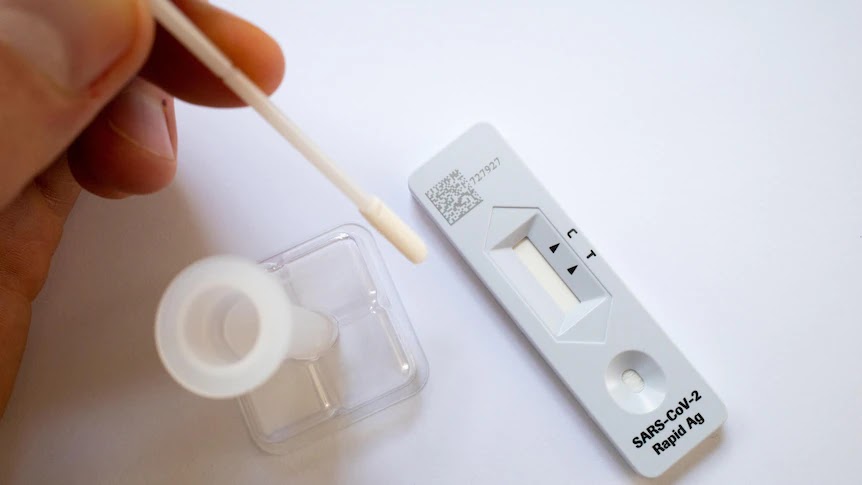
Several Australian firms have been waiting months for local approval of their COVID-19 fast antigen tests, as Australians struggle to obtain one. Only one of the 22 home tests approved by Australia’s Therapeutic Goods Administration (TGA) is manufactured in Australia, with the rest coming from China, the United States, Korea, Singapore, and Germany. There are concerns that if planned cargoes are diverted elsewhere, Australia’s present supply crisis may worsen. AnteoTech has obtained some government financing, including a $1.4 million promise from the Queensland government in early 2021 and a roughly $2 million rebate under the federal R&D Tax Incentive Scheme.
Several Australian firms have developed COVID RATs, but at least two are awaiting TGA certification for tests that are currently in use in Europe or North America. Ellume, another Brisbane-based firm, is in the process of obtaining regulatory certification for its COVID RAT. Lumos Diagnostics’ executive chairman, Sam Lanyon, said his company has pushed the Australian government to enable domestic RAT production as soon as mid-2020. He adds that he was amazed at how quickly it changed based on the pandemic’s demands and Delta’s coming in and driving a whole lot of legislative changes that he thinks would have been wonderful. He told ABC News that his company addressed the federal and state governments about a year and a half ago, urging them to support local manufacturing of COVID RATs.
According to CEO David Thomson, the Australian application for Anteo Tech corresponded with stricter evidentiary criteria than when his test was approved for use in Europe. AnteoTech and Lumos Diagnostics both submitted applications in September, and both have been invited to provide additional information. The federal Department of Health stated in a statement that “approval can take anywhere from 3–5 days if all relevant paperwork is submitted.” Although rapid antigen tests are more prone than PCR testing to producing false negatives, experts say they are valuable for determining when a person is at their most contagious. As soon as information from applicants is received, the TGA will continue to “prioritize and expedite” the examination of COVID-19 fast antigen self-tests for home use.
“We haven’t encountered anyone who has gone through in three to five days,” said Derek Thomson, the company’s CEO. AnteoTech said it planned to submit further new data to the agency next week, but that it did not expect speedy approval. Following a decision by Australia’s national cabinet to eliminate the requirement for PCR testing, the country will no longer be able to furnish accurate COVID-19 case statistics. On a blue surface, a Rapid Antigen Test packet, a swab, a dropper bottle, and the test itself are laid out. Some medical specialists are concerned that Australia’s statistics would lose their accuracy, while others are less concerned.